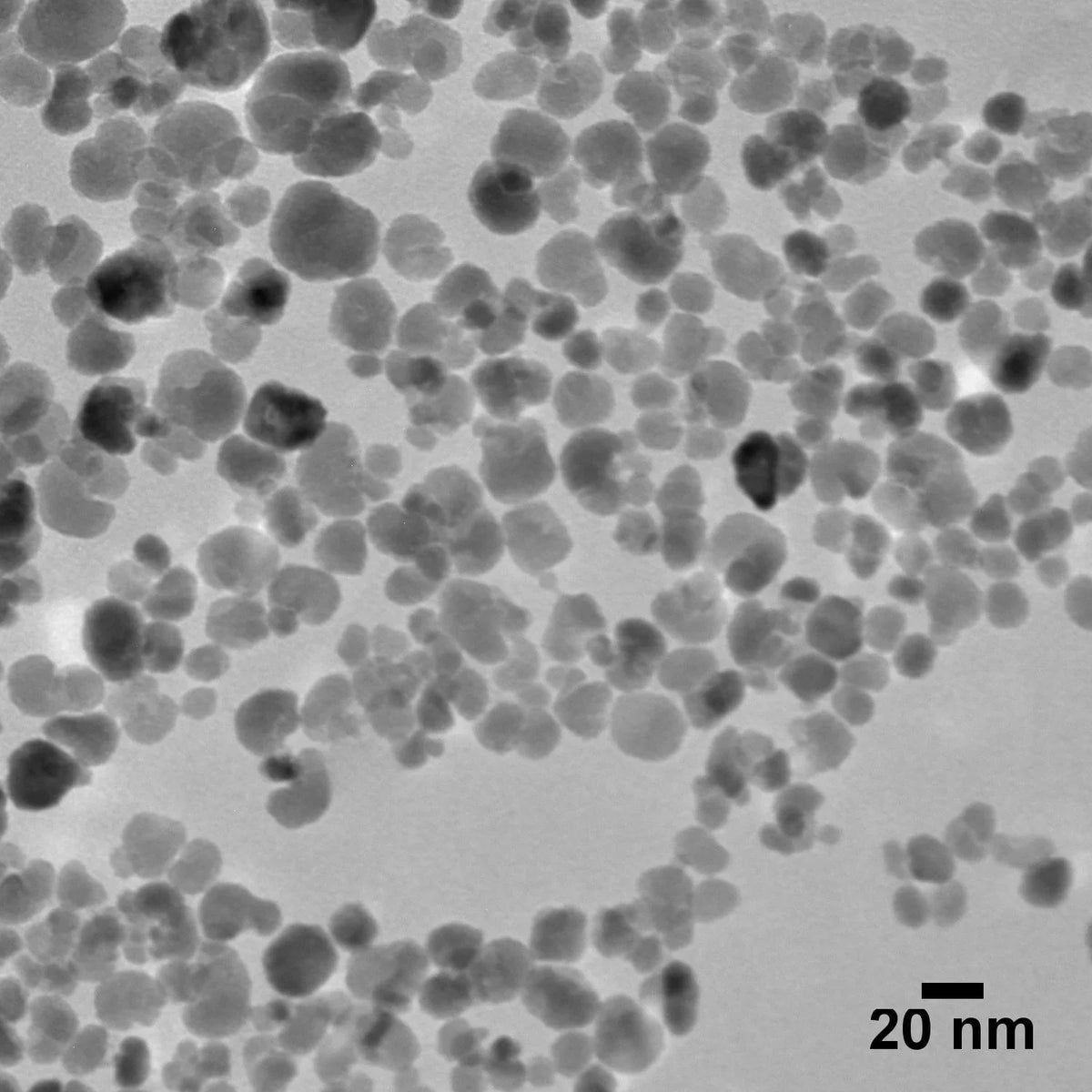
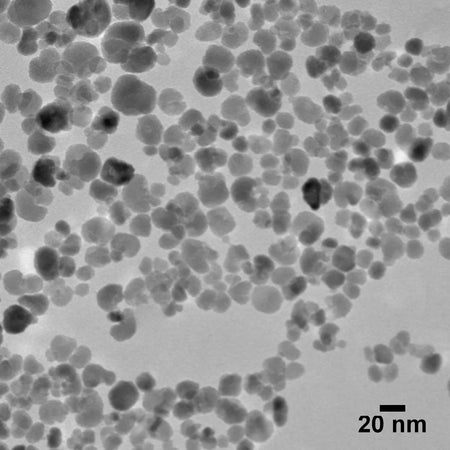
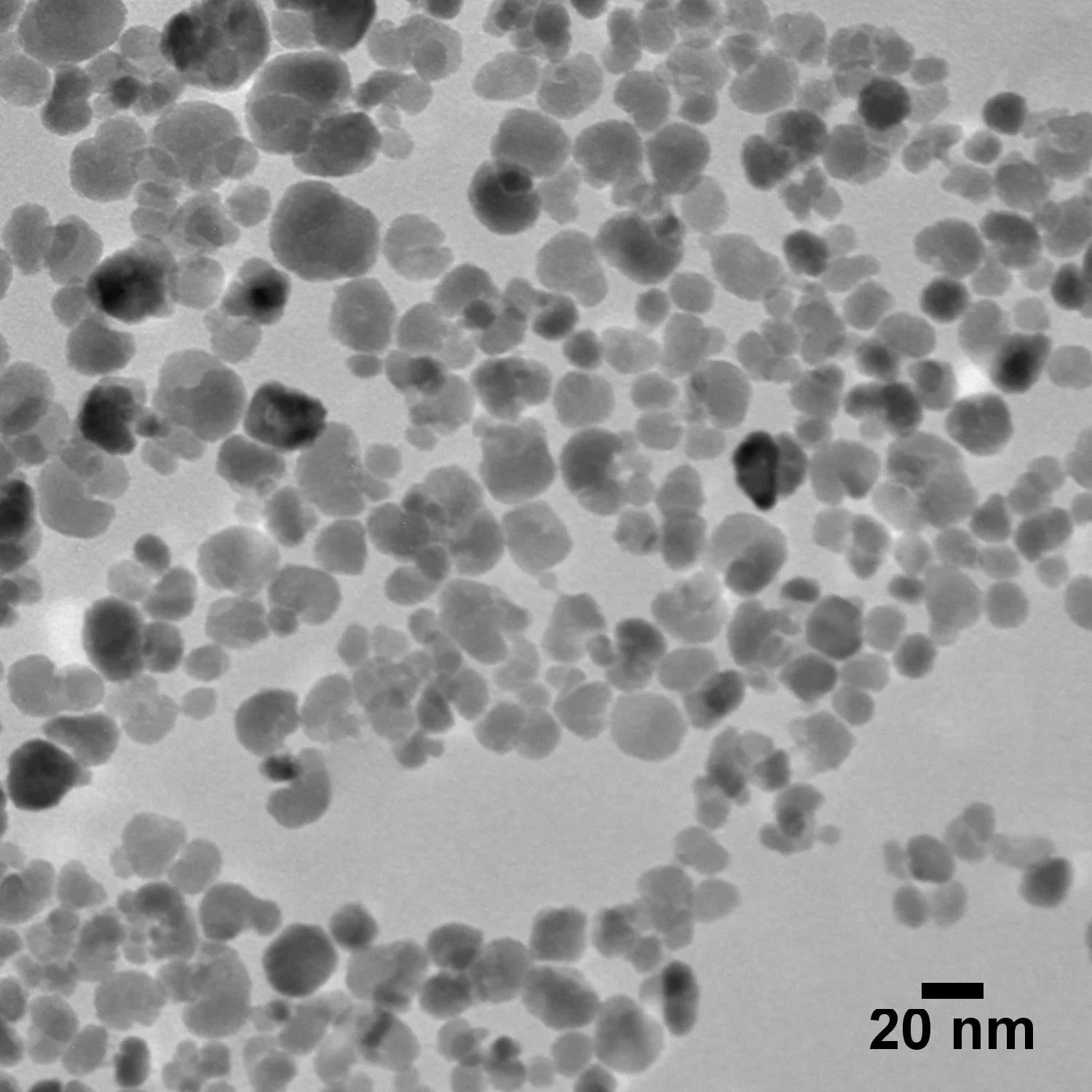
20 nm Magnetite Nanoparticles
Customers outside of the US & Canada: please visit your local distributor’s website to purchase this item using its SKU, or contact us here. Thank you!
nanoComposix specializes in providing high-purity and monodisperse nanomaterials – delivering the quality you need for consistent results.
20 nm magnetite (iron oxide) nanoparticles combine superparamagnetic properties at ambient temperatures with a high surface area to volume ratio. These nanoparticles are useful in a broad range of applications from nanomedicine, imaging contrast agents, drug delivery, hyperthermia therapy, biosensors, and more.
nanoComposix provides high quality nanomaterials that are precisely engineered and highly characterized. All our products are supplied with a batch-specific Certificate of Analysis including characterization data such as TEM, DLS, Zeta, and UV-Vis.
Intended for Research Use Only. Not for use in diagnostic procedures. Contact us for custom formulations or click below for bulk supply requirements.
| Overview | |
|---|---|
| Surface | PVP |
| Description | Large polymer surface. Stable in a wide variety of solvents. |
| Solvent | 2 mM sodium citrate |
| Surface chemistry | PVP 40 kDa |
| Specifications | |
| Surface | PVP |
| TEM Diameter | 20 ± 5 nm |
| CV | ≤ 30% |
| Documents | |
| Surface | PVP |
| Example Certificate of Analysis (CoA) | Download |
| Safety Data Sheet (SDS) | 20 mg/mL (NanoXact) |
| Storage & Handling | Download |
| Expected Ranges | |
| Surface | PVP |
| Hydrodynamic diameter (DLS) | TEM diameter + 20–45 nm |
| Zeta potential | ≤ –30 mV |
| pH of solution | 6.0–7.0 |